Since the establishment of the principles of relativity, the problem arose of determining the transformations that would allow the expression of thermodynamic parameters in the relativistic context, such as entropy, temperature, pressure or heat transfer; in a analogous way to that achieved in the context of mechanics, such as space-time and momentum.
The first efforts in this field were made by Planck [1] and Einstein [2], arriving at the expressions:
S’ = S, T’ = T/γ, p = p’, γ = (1 – (v/c)2) -1/2
Where S, T, p are the entropy, temperature and pressure of the inertial thermodynamic system at rest I, and S’, T’, p’ are the entropy, temperature and pressure observed from the inertial system I’ in motion, with velocity v.
But in the 1960s this conception of relativistic thermodynamics was revised, and two different points of view were put forward. On the one hand, Ott [3] and Arzeliès [4] propose that the observed temperature of a body in motion must be T’ = Tγ. Subsequently, Landsberg [5] proposes that the observed temperature has be T’ = T.
All these cases are based on purely thermodynamic arguments of energy transfer by heat and work, such that ∆E = ∆Q + ∆W. However, van Kampen [6] and later Israel [7] analyze the problem from a relativistic point of view, such that ∆G = ∆Q + ∆W , where ∆G is the increment of the energy-momentum vector, ∆Q and ∆W are the four-component vectors corresponding to the irreversible and reversible part of the thermodynamic process, with ∆Q being the time component of ∆Q .
Thus, the van Kampen-Israel model can be considered the basis for the development of thermodynamics in a relativistic context, offering the advantage that it does not require the concepts of heat and energy, the laws of thermodynamics being expressed in terms of the relativistic concept of momentum-energy.
In spite of this, there is no theoretical justification based on any of the models that allows to determine conclusively the relation of temperatures corresponding to the thermodynamic system at rest and the one observed in the system in motion, so that the controversy raised by the different models is still unresolved today.
To complicate the situation further, the experimental determination of the observed temperature poses a challenge of enormous difficulty. The problem is that the observer must move in the thermal bath located in the inertial system at rest. To find out the observed temperature from the moving reference system Landsberg proposed a thought experiment and thus determine the relativistic transformation of the temperature experimentally. As a result of this proposal, he recognized that the measurement scenario may be unfeasible in practice.
In recent years, algorithms and computational capabilities have made it possible to propose numerical solutions aimed at resolving the controversy over relativistic transformations for a thermodynamic system. As a result, it is concluded that any of the temperature relations proposed by the different models can be true, depending on the thermodynamic assumptions used in the simulation [8] [9], so the resolution of the problem remains open.
The relativistic thermodynamic scenario
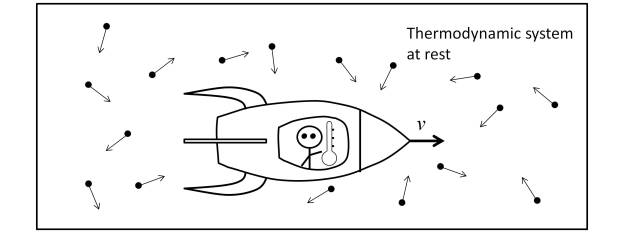
In order to highlight the difficulty inherent in the measurement of the thermodynamic body temperature of the inertial system at rest I from the inertial system I’ it is necessary to analyze the measurement scenario.
Thus, as Landsberg and Johns [10] make clear, the determination of the temperature transformation must be made by means of a thermometer attached to the observer by means of a brief interaction with the black body under measurement. To ensure that there is no energy loss, the observer must move within the thermodynamic system under measurement, as shown in the figure below.
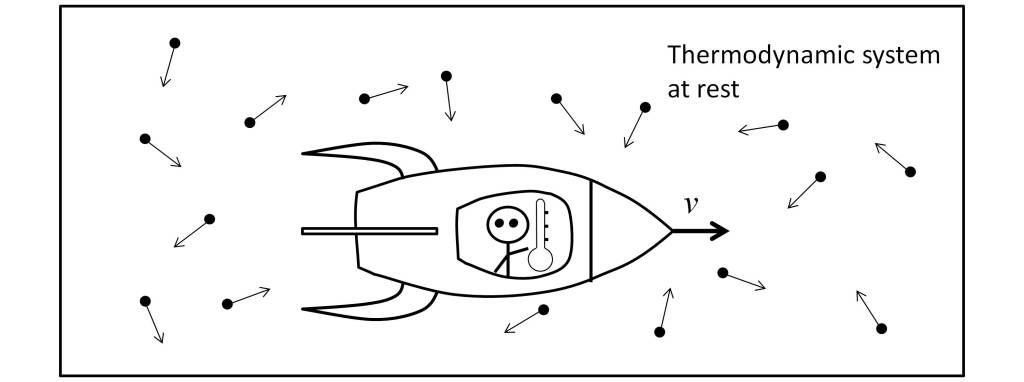
This scenario, which may seem bizarre and may not be realizable in practice, clearly shows the essence of thermodynamic relativity. But it should not be confused with the scenario of temperature measurement in a cosmological scenario, in which the observer does not move inside the object under measurement.
Thus, in the case of the measurement of the surface temperature of a star the observer does not move within the thermodynamic context of the star, so that the temperature may be determined using Wien’s law, which relates the wavelength of the emission maximum of a black body and its temperature T, such that T = b/λmax, where b is a constant (b≅2.9*10-3 m K).
In this case the measured wavelength λmax must be corrected for several factors, https://en.wikipedia.org/wiki/Redshift, such as:
- The redshift or blueshift produced by the Doppler effect, a consequence of the relative velocity of the reference systems of the star and the observer.
- The redshift produced by the expansion of the universe and which is a function of the scales of space at the time of emission and observation of the photon.
- The redshift produced by the gravitational effect of the mass of the star.
As an example, the following figure shows the concept of the redshift produced by the expansion of the universe.

Entropy is a relativistic invariant
Although the problem concerning the determination of the observed temperature in relativistic systems I and I’ remains open, it follows from the models proposed by Planck, Ott and Landsberg that the entropy is a relativistic invariant.
This conclusion follows from the fact that the number of microstates in the two systems is identical, so that according to the expression of the entropy S = k ln(Ω), where k is the Boltzmann constant and Ω is the number of microstates, it follows that S = S’, since Ω = Ω’.
The invariance of entropy in the relativistic context is of great significance, since it means that the amount of information needed to describe any scenario of reality that emerges from quantum reality is an invariant, independently of the observer.
In the post ’an interpretation of the collapse of the wave function‘ it was concluded that a quantum system is reversible and therefore its entropy is constant and, consequently, the amount of information for its description is an invariant. This post also highlights the entropy increase of classical systems, which is deduced from the ‘Pauli’s Master Equation’ [11], such that Ṡ > 0. This means that the information to describe the system grows systematically.
The conclusion drawn from the analysis of relativistic thermodynamics is that the entropy of a classical system is the same regardless of the observer and, therefore, the information needed to describe the system is also independent of the observer.
Obviously, the entropy increment of a classical system and how the information increment of the system emerges from quantum reality remains a mystery. However, the fact that the amount of information needed to describe a system is an invariant independent of the observer suggests that information is a fundamental physical entity at this level of reality.
On the other hand, the description of a system at any level of reality requires information in a magnitude that according to Algorithmic Information Theory is the entropy of the system. Therefore, reality and information are two entities intimately united from a logical point of view.
In short, from both the physical and the logical point of view, information is a fundamental entity. However, the axiomatic structure that configures the functionality from which the natural laws emerge, which determines how information is processed, remains a mystery.
| [1] | M. Planck, « Zur dynamik bewegter systeme,» Ann. Phys. , vol. 26, 1908. |
| [2] | A. Einstein, «Über das Relativitätsprinzip und die aus demselben gezogenen Folgerungen.,» Jahrb. Radioakt. Elektron., vol. 4, pp. 411-462, 1907. |
| [3] | H. Ott, «Lorentz-Transformation der Wiirme und der Temperatur,» Zeitschrift Für Physik, vol. 175, pp. 70-104, 1963. |
| [4] | H. Arzeliès, «Transformation relativiste de la temperature et de quelques autres grandeurs thermodynamiques.,» Nuovo Cim. 35, 792 (1965)., vol. 35, nº 3, pp. 792-804, 1965. |
| [5] | P. Landsberg, «Special relativistic thermodynamics,» Proc. Phys. Soc., vol. 89, pp. 1007-1016, 1966. |
| [6] | N. G. van Kampen, «Relativistic Thermodynamics of Moving Systems,» Phys. Rev. 173, 295 (1968)., vol. 173, pp. 295-301, 1968. |
| [7] | W. Israel, «Nonstationary Irreversible Thermodynamics: A Causal Relativistic Theory,» Ann. Phys., vol. 106, pp. 310- 331, 1976. |
| [8] | D. Cubero, J. Casado-Pascual, J. Dunkel, P. Talkner y P. Hänggi, «Thermal Equilibrium and Statistical Thermometers in Special Relativity.,» Relativity. Phys. Rev. Lett. , vol. 99, pp. 170601, 1-4, 2007. |
| [9] | R. Manfred, «Thermodynamics meets Special Relativity–or what is real in Physics?,» arXiv: 0801.2639., nº 2008. |
| [10] | P. T. Landsberg y K. A. Johns, «The Problem of Moving Thermometers,» Proc. R. Soc. Lond., vol. 306, pp. 477-486, 1968. |
| [11] | F. Schwabl, Statistical Mechanics, pp. 491-494, Springer, 2006. |